ACCESS Model Overview
Overview
The Centers for Medicare & Medicaid Services (CMS) Innovation Center (CMMI) has launched a new demonstration model, the Advancing Chronic Care with Effective, Scalable Solutions (ACCESS) Model. This voluntary model will be a 10-year national test of a payment approach focused on the role of technology in care delivery. ACCESS tests a new payment approach, using Outcome-Aligned Payments (OAPs) for technology-enabled care delivery and management of certain chronic conditions in Medicare fee-for-service (FFS). ACCESS is designed to expand access to technology-enabled, longitudinal chronic care by replacing visit-based fee-for-service payment with predictable, prospective per-beneficiary payments tied to measurable clinical and patient-reported outcomes, rather than volume of services. According to CMS, this model is intended to address payment barriers in Medicare FFS payment systems that make it challenging to leverage technologies to deliver continuous, remote, and outcomes-driven chronic disease management, while still maintaining accountability for care outcomes.
Model Scope and Application Timeline
The model will operate nationwide from July 5, 2026 through June 30, 2036, with rolling application cycles. Applications for the first cohort will open on January 12, 2026, and are due April 1, 2026, with subsequent cohorts beginning quarterly through July 1, 2033. Applications will be accepted on a rolling basis through April 1, 2033 to enable at least two years of participation for each participant.
Model Timeline
| Milestone | Date |
|---|---|
| Applications Open | January 12, 2026 |
| Initial Application Deadline | April 1, 2026 |
| First Cohort Begins | July 5, 2026 |
| Subsequent Cohorts Begin | January 1, 2027, and quarterly thereafter through July 1, 2033 |
| Applications Close | April 1, 2033 |
| Model Performance Period Ends | June 30, 2036 |
See Table 2. Model Timeline, ACCESS RFA
Model Eligibility and Participation
Participation is defined at the organizational level and identified by a single Medicare Part B –enrolled TIN that is eligible to bill under the Medicare Physician Fee Schedule (PFS). Eligible entities include Medicare Part B–enrolled providers and suppliers, excluding DMEPOS suppliers and laboratory suppliers. Organizations not enrolled in Part B must complete enrollment before participation is fully approved.
In addition, ACCESS participants must:
- maintain active Medicare Part B enrollment under a single TIN;
- ensure that all physicians and non-physician practitioners who furnish or supervise care under the model are individually enrolled in Medicare as participating providers or suppliers, have reassigned their Medicare billing rights to the participating organization’s TIN, and are licensed and practicing within applicable state scope-of-practice requirements;
- submit and maintain an up-to-date roster of all Medicare-enrolled practitioners (with NPIs) furnishing or supervising care under the TIN; and
- successfully complete CMS program integrity screening and any other reviews conducted under Medicare’s Conditions for Medicare Payment as described in 42 C.F.R. Part 424.
Medical Director Requirements

Each participant must also designate a Medicare-enrolled physician as medical director, responsible for clinical oversight, patient safety, outcomes performance, and regulatory compliance. A participant may contract with a self-employed physician or physician employed by a professional entity or physicians group for the Medical Director role, provided the contract specifies the physician who assumes Medical Director responsibilities and obligations. Notably, this requirement is closely modeled on CMS medical director requirements used in hospice (42 CFR § 418.102) and End-Stage Renal Disease (ESRD) facilities (42 CFR § 494.150).
Participation Requirements
To participate in the model, ACCESS participants must also, among other things, submit ACCESS services using model-specific G-codes, submit outcome data to CMS via a FHIR-based application programming interface (API), meet care coordination and health IT requirements, and comply with all applicable federal and state laws and requirements, including, but not limited to, licensure and scope of practice, HIPAA, and applicable U.S. Food and Drug Administration (FDA) requirements for technologies treated as medical devices.
Program Integrity Screening
CMS indicates it will conduct program integrity screening of applicants and may deny or terminate participation based on screening results; it also describes ongoing monitoring and audits tied to outcomes, utilization, billing, and data validation.
Beneficiary Eligibility and Alignment
ACCESS is designed for Original Medicare beneficiaries with qualifying chronic conditions who voluntarily and prospectively align to an ACCESS participant. Beneficiaries may voluntarily and prospectively align to an ACCESS participant by track. Enrollment does not restrict beneficiary freedom of choice for other Medicare-covered services, and beneficiaries may enroll in multiple tracks concurrently (with limited exceptions). Alignment does not restrict freedom of choice for other covered Original Medicare services for Medicare beneficiaries.
A beneficiary must meet:
- clinical eligibility with track-specific qualifying conditions below;
- be enrolled in Original Medicare Parts A and B, with Medicare as the primary payer; and
- not be enrolled in Medicare Advantage, PACE, or the Medicare hospice benefit.
Beneficiaries may align to only one ACCESS participant per track at a time, but may align to multiple tracks (same or different participants), except they may not be enrolled in both eCKM and CKM simultaneously because CKM encompasses eCKM measure and care.
Participants must obtain and document informed consent (verbal or written permitted) and explain key terms, including that switching/disenrollment is permitted after 90 days from the start of alignment.
Clinical Tracks and OAP Measures
Initially, ACCESS will begin with four clinical participation tracks based on chronic conditions that are considered suitable for technology-enabled care:
- Early Cardio-Kidney-Metabolic (eCKM)
- Cardio-Kidney-Metabolic (CKM)
- Musculoskeletal (MSK)
- Behavioral Health (BH)
| Track | Qualifying Conditions [3,4] | OAP Measure |
|---|---|---|
| eCKM | Hypertension (HTN), or two or more of the following conditions: dyslipidemia, obesity or overweight with marker of central obesity, prediabetes |
|
| CKM | One or more of the following conditions: diabetes mellitus, chronic kidney disease (CKD),7 atherosclerotic cardiovascular disease (ASCVD) |
|
| MSK | Chronic musculoskeletal (MSK) pain |
|
| BH | One or more of the following conditions: depression, anxiety |
|
See Table 1. Initial Tracks Overview, ACCESS RFA
Additional clinical tracks and conditions may be considered in future years.
Care Delivery
ACCESS is structured to hold participants accountable for outcomes while also providing flexibility to deliver integrated services through modalities that can include in-person, virtual, asynchronous, or other technology-enabled approaches when clinically appropriate and lawful.
CMS provides non-exhaustive examples of services that may be delivered under ACCESS, including:
- Clinician consultations
- Lifestyle support services such as nutrition counseling, exercise support, and smoking cessation interventions • Remote monitoring via connected medical devices
- Therapy and behavioral health counseling
- Patient education and care coordination
- Medication prescription and management
- Ordering and interpretation of laboratory and diagnostic tests
- Connected device deployment and monitoring
- Deployment and oversight of FDA-cleared, authorized, or approved software devices
In addition, ACCESS participants must:

- Deliver care reasonably designed to be safe, high-quality, and consistent with nationally recognized clinical guidelines and standards of practice.
- Identify when needs exceed ACCESS scope and facilitate transitions to appropriate clinicians and settings to support continuity and patient safety.
CMS indicates it will monitor claims and clinical submissions for quality and patient safety, including patterns suggesting overtreatment, and may take corrective action or remove participants with persistent issues.
Payment Design
OAPs are standard per-patient payments for managing all qualifying conditions a beneficiary has within a track over 12-month care periods, and are designed to shift incentives from volume to outcomes. OAPs do not include the costs of medications, labs, imaging, or DMEPOS, which may be coordinated but are billed separately through Medicare FFS by financially unaffiliated entities.
Payment Tiers
ACCESS has two payment tiers:
- Initial Period (12 months): higher rate for onboarding and initial clinical improvement; which generally applies when first treating the beneficiary in the track within the past two years and at least one required measure is not at target.
- Follow-On Period: lower rate for ongoing management and maintenance when measures are already controlled at baseline or the participant treated the beneficiary in the same track within the past two years.
Claim Submission
Participants submit monthly claims using new track-specific G-codes to Medicare Administrative Contractors (MACs). Claims are processed as zero-paid by the MACs and then quarterly payments are issued by the Innovation Payment Contractor (IPC) based on validated claims.
Split Payments
Through the IPC, CMS pays up to 50% of the annual OAP amount during the care period, via quarterly installments for validated monthly claims. The remaining 50% is withheld and reconciled after the 12-month care period concludes, which is further adjusted based on performance.
Performance Adjustments
OAP payments may be adjusted downward based on the following:
- Clinical Outcome Adjustment: This adjustment is based on an Outcome Attainment Rate (5) (OAR) compared to an Outcome Attainment Threshold (OAT), which reflects the minimum OAR needed to earn full payment. For the first performance year, the OAT will be 50 percent. If OAR is below the OAT, payment is reduced proportionally, but the adjustment is capped at a 50% reduction to the full OAP amount.
- Substitute Spend Adjustment: This adjustment is based on a Substitute Spend Rate (SSR) (6) compared to a Substitute Spend Threshold (SST), which reflects the minimum SSR needed to earn full payment. For the first performance year, the SSR will be 90 percent. If the SSR is below the SST, payment is reduced proportionally, but the adjustment is capped at a 25% reduction to the full OAP amount. Each clinical track includes a Substitute Spend List (7) which identifies which services are considered substitutes when delivered by another Medicare entity for the same condition.
CMS will reconcile semi-annually and apply only the larger of the two adjustments in a reconciliation period to avoid compounding penalties.
Co-Management Payments
To support integration with beneficiaries’ existing care teams, CMS will provide a billable new ACCESS Co-Management service that eligible clinicians may bill for documented review of ACCESS updates and care coordination. This will be paid at approximately $30 per service, (8) limited to once every four months per beneficiary per track, with no Part B cost-sharing. A one-time modifier for onboarding support may add approximately $10 the first time billed for a beneficiary.
Cost Sharing
CMS indicates it will offer a patient incentive safe harbor (42 CFR 1001.952(ii)(2)) to ACCESS participants to waive beneficiary cost-sharing for OAPs as a beneficiary engagement incentive. This policy must be uniformly applied to all beneficiaries. To the extent a participant intends to collect beneficiary cost sharing of OAP payments, this along with the expected beneficiary payment amount must be clearly disclosed before beneficiary enrollment.
Data and Care Reporting
Participants check eligibility via an Eligibility API, align beneficiaries via an Alignment API, submit outcomes via a FHIR-based Reporting API, and may request claims data via the BCDA API to support care coordination and monitoring. ACCESS participants are not required to use Certified EHR Technology, but must meet minimum HIT expectations aligned with ONC standards, including:
- standardized APIs for patient/population services (referencing ONC certification criteria at 45 CFR § 170.315(g)(10) and USCDI);
- connectivity to a health information exchange (HIE) enabling bidirectional exchange within 12 months of model start; and
- ability to submit required outcomes to CMS’s FHIR server.
Care Coordination
Participants must make reasonable efforts to identify the beneficiary’s primary care provider (PCP) or referring clinician and, with beneficiary consent, send standardized electronic updates at certain timeframes:
- Care initiation: within 10 days of initiation.
- Care completion: within 30 days after the end of the 12-month period, or sooner if the beneficiary disenrolls or transitions care.
- Care escalation: within 10 days of a transition to another clinician or care setting when beneficiary clinical needs exceed the ACCESS services’ scope.
CMS will provide a standardized care plan update template, and participants must document attempts and outcomes of transmission efforts.
ACCESS Participant Flow
CMS has created a flowchart explaining the ACCESS beneficiary onboarding and payment process:
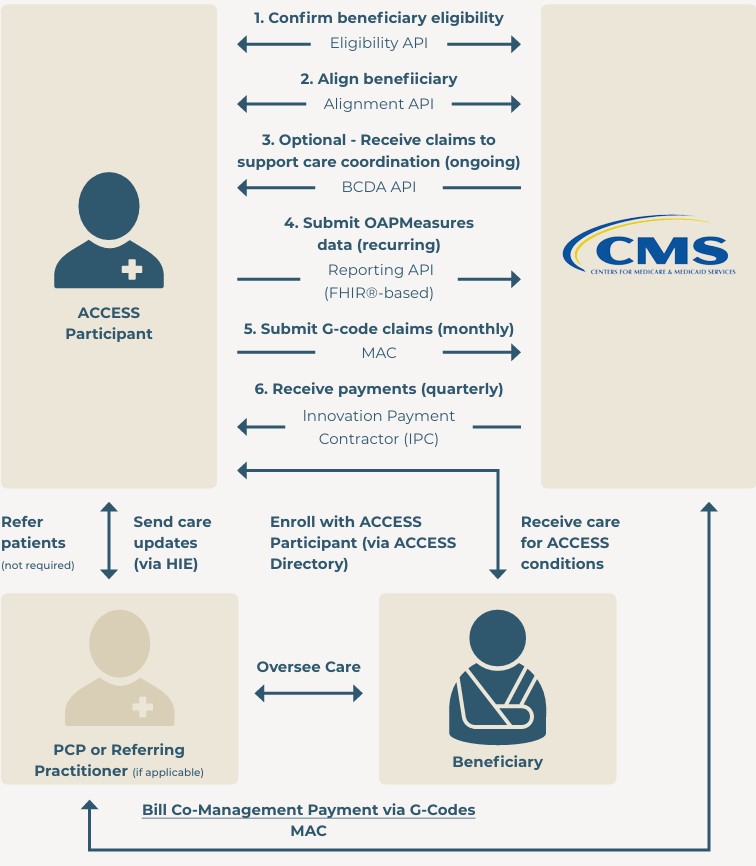
See Figure 1. ACCESS Participant End-to-End Flow, ACCESS RFA
Footnotes
1. A beneficiary must not have a clinically excluded condition, as outlined within the Required Clinical Exclusions list in Appendix C of the Request for Applications.
2 Refer to the RFA for additional details about qualifying conditions.
3. A beneficiary must not have a clinically excluded condition, as outlined within the Required Clinical Exclusions list in Appendix C of the Request for Applications.
4. Refer to the RFA for additional details about qualifying conditions.
5. The OAT reflects the percentage of aligned beneficiaries who complete their 12-month period meet all required OAP measure targets.
6. The SSR reflects the percentage of aligned beneficiaries who did not receive listed substitute services from other Medicare providers or suppliers for the same condition during their ACCESS care period.
7. See Appendix E of the ACCESS RFA.
8. This may be further adjusted based on geography or other adjustments in the Medicare Physician Fee Schedule.
Want to Print or Share?
Download the PDF.